
- Видео 224
- Просмотров 146 952 891
Tyler DeWitt
США
Добавлен 22 мар 2007
Need help with Chemistry? Whether you're in high school, college, AP or IB courses, these videos can help! They are also targeted to students studying for the AP Test, SAT, MCAT, DAT, and OAT exams, as well as students in nursing, nutrition, pre-med and pre-vet programs. We'll cover the introductory topics patiently and clearly, using lots of examples, analogies and practice problems. The main topics covered in this library are: Significant Figures, Scientific Notation, Density, Stoichiometry, Nuclear Chemistry, VSEPR Theory, Atomic Structure, Covalent and Ionic Bonding, Thermochemistry, and Gases.
Mole Fraction Introduction
This video is an introduction to mole fraction. Mole fraction is one of the ways that we can describe or measure the concentration of different components in a mixture. It is calculated by dividing the moles of the component of interest by the total number of moles in the mixture. This video is for students in both high school and college/university. It will also be helpful to people who are preparing for the following curricula and exams: AP, SAT, IB, A-levels, O-levels, IGCSE, ICSE, CBSE, MCAT, DAT, OAT, JEE, NEET
Просмотров: 11 551
Видео
Molarity: A Deeper Understanding
Просмотров 18 тыс.3 месяца назад
Most students can work through the math formula for molarity (M), but they don't really understand what they're doing, or why they're doing. In many cases, students can't really explain what molarity is or what it means. Molarity is defined as moles per liter, and this information is very helpful actually understanding how to rearrange the equation, and solve problems in a way that makes sense....
Mole Fraction Practice Problems
Просмотров 9 тыс.3 месяца назад
Here, we work through two practice problems involving mole fraction. Mole fraction is calculation that is used to determine the concentration of components in a mixture. The number of moles of a certain component is divided by the total number of moles in the mixture. When the numbers are not given in moles, you first have to convert from gams to moles by using the molar mass. This video will b...
TEAS 7 Chemistry: Electrons and Ions
Просмотров 16 тыс.5 месяцев назад
TEAS Chemistry in One Day Full Course: www.teasinoneday.com This video for the TEAS Chemistry section covers electrons and ions. We look at how the positive charges of protons and the negative charges of electrons can cancel each other out, creating a net charge for an atom. We learn how to determine the number of protons and electrons in ions with different net charges, and what creates an ato...
TEAS 7 Chemistry: Protons and Atomic Number
Просмотров 11 тыс.7 месяцев назад
For my full course on the Chemistry section for the ATI TEAS 7: www.teasinoneday.com/welcome This video is specifically for people who are preparing the ATI TEAS 7 exam. We cover all of the Chemistry that you'll need to know for the TEAS exam. Here, we talk about protons and atomic number, which are two very important things for understanding atoms. The number of protons or the atomic number de...
AP Chemistry: How to Work Smarter, Not Harder
Просмотров 14 тыс.7 месяцев назад
For free AP® Chemistry resources: www.tdwscience.com/apchem AP Chemistry can be very challenging, but students often take the wrong approach. They take a brute force approach, give up hours of sleep, and practically cry blood. Instead, the AP Chemistry exam rewards students who take an approach that is "smarter, not harder." Students tend to do best on the AP Chemistry exam when the work to und...
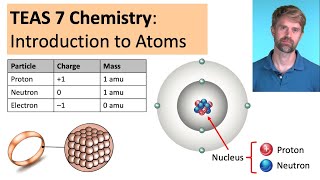
TEAS 7 Chemistry: Introduction to Atoms
Просмотров 18 тыс.7 месяцев назад
Check out the full TEAS Chemistry course and resources at: www.teasinoneday.com/welcome This is a video about atoms and atomic structure that is specifically for people who are taking the TEAS exam. We cover all of the relevant information that is most important for the TEAS 7 Chemistry section, such as atoms, elements, and subatomic particles (protons, neutrons, and electrons) along with their...
Introduction to the AP Chemistry Multiple Choice Questions (MCQ's)
Просмотров 20 тыс.9 месяцев назад
Students often say that the multiple choice questions (MCQ's) are the hardest part of the AP Chemistry test. And they really are tricky. The MCQ's are different from the sort of multiple choice questions that show up on most other types of exams, and they test multiple concepts at the same time. They also require some math skills and puzzle solving. Here, we work through a bunch of AP Chemistry...
Ionization Energy and the Photoelectric Effect (AP Chemistry)
Просмотров 10 тыс.11 месяцев назад
This is an AP Chemistry multiple choice question (MCQ) focusing on ionization energy and the photoelectric effect. During the photoelectron effect, a photo hits an atom and ejects an electron. Based on the kinetic energy of the electron, it possible to determine characteristics about the electron's location. The question also asks about electron orbitals, shielding, kinetic energy, ionization e...
Acid Conjugate Base Multiple Choice Question (AP Chemistry)
Просмотров 12 тыс.11 месяцев назад
Acids and conjugate bases classify compounds based on whether they accept or donor protons (H ions). Acids donate or give up a proton or H ion, and bases accept a proton of H ion. This classification is known as the Bronsted Lowry theory of acids and bases. Compounds can be classified as acids and conjugate bases, as well as bases and conjugate acids. Often, we need to identify the acid/conjuga...
Delta G, Delta H, and Delta S Problem (AP Chemistry)
Просмотров 7 тыс.11 месяцев назад
Delta G (Gibbs Free Energy), Delta H (Enthalpy), and Delta S (Entropy) define whether a reaction will be thermodynamically favorable or thermodynamically unfavorable. These terms are often also referred to as "spontaneous" or "non-spontaneous." This is a multiple choice question (MCQ) that is specifically designed to prepare for the AP Chemistry test, but may also be relevant for other exams an...
Entropy and Reactions Problem (AP Chemistry)
Просмотров 7 тыс.Год назад
This is an AP Chemistry-style multiple choice question (MCQ) focusing on entropy (delta S) and reactions. We have to determine which reaction illustrates the least increase in entropy. Entropy measures the amount of disorder in a system. In order to figure this problem out, we need to keep two essential entropy trends in mind: entropy increases as we transition from solid to liquid to gas. Entr...
Dilutions and Lab Procedure (AP Chemistry)
Просмотров 6 тыс.Год назад
This AP Chemistry multiple choice question (MCQ) video focuses on dilutions and lab procedure. These are common topics on the AP Chemistry exam. First, you need to dilute a stock solution to produce a working solution. This requires using the dilution equation, M1V1 = M2V2. There are common mistakes that students often make, and the answer choices are designed to try to trick and trap you. Then...
Kinetics and Reaction Rates (AP Chemistry)
Просмотров 8 тыс.Год назад
Here, we work through an AP Chemistry multiple choice question (MCQ) that focuses on kinetics and reaction rates. Specifically, we look at the method of initial rates, where we look at how the initial concentration of a reactant changes, and see what effect that has on the rate of the reaction. There are a number of different ways to solve a problem like this: we will look at a logic-based appr...
Solubility Curves and Practice Problems
Просмотров 67 тыс.Год назад
Here, we look at solubility curves. We see what they mean, how to read them, and how to answer questions using them. We begin with an overview of solutes, solvents, solutions, and solubility, and then look at general trends for solubility and temperature. Then, we look at the process for reading a solubility curve to determine solubilities and different temperatures, and vice versa. Then, we lo...
Electroplating Calculations (AP Chemistry)
Просмотров 6 тыс.Год назад
Electroplating Calculations (AP Chemistry)
Intermolecular Forces and Boiling Point (AP Chemistry)
Просмотров 27 тыс.Год назад
Intermolecular Forces and Boiling Point (AP Chemistry)
States of Matter (Phases of Matter): Solids, Liquids, and Gases
Просмотров 45 тыс.Год назад
States of Matter (Phases of Matter): Solids, Liquids, and Gases
Calculating Molarity (Silent Movie Style)
Просмотров 33 тыс.Год назад
Calculating Molarity (Silent Movie Style)
Radioactive Decay Calculations Practice Problem
Просмотров 37 тыс.Год назад
Radioactive Decay Calculations Practice Problem
AP® Chemistry: Bonding, Hybridization, Intermolecular Forces, Enthalpy
Просмотров 81 тыс.2 года назад
AP® Chemistry: Bonding, Hybridization, Intermolecular Forces, Enthalpy
AP® Chemistry Kinetics Questions Free Response
Просмотров 27 тыс.2 года назад
AP® Chemistry Kinetics Questions Free Response
AP® Chemistry Weak Acid Equilibrium Questions
Просмотров 39 тыс.2 года назад
AP® Chemistry Weak Acid Equilibrium Questions
AP® Chemistry Multiple Choice Practice Problems
Просмотров 111 тыс.2 года назад
AP® Chemistry Multiple Choice Practice Problems
Unsaturated, Saturated, and Supersaturated Solutions
Просмотров 176 тыс.3 года назад
Unsaturated, Saturated, and Supersaturated Solutions
Rate of Dissolving and Factors that Affect It
Просмотров 74 тыс.3 года назад
Rate of Dissolving and Factors that Affect It
Pressure and Gas Solubility (Henry's Law)
Просмотров 78 тыс.3 года назад
Pressure and Gas Solubility (Henry's Law)
Temperature and Solubility: Solids and Gases
Просмотров 100 тыс.3 года назад
Temperature and Solubility: Solids and Gases








Tyler is an excellent teacher, who can stretch out his imagination to explain abstract scientific concepts in a very simple manner. Thank you Tyler ; you are an asset to students who are learning from RUclips.
you changed my life brother
I learned this 31 years ago, knowing all the rules enough to get an 'A', yet I dont know why until now. LOL!!!!!! Thanks Teach!
If only someone had broken it down and explained it to Me when I was a kid I would have LOVED chemistry! THANK YOUYOU!!!
@tdewitt451 Thank you hardly touches what needs to be said to you. As a teacher, I commend you for taking the time to go through every single step of figuring out each of these and explaining it over and over so very clearly. I was struggling until I watched your video - now it all works. It's surprising to me how simple this concept is, but how cloudy other teachers can make it. I appreciate your clarity and not making it more difficult than it needs to be. I hope I can teach it as well as you do when I get up in front of one of my first chemistry classes! Thankfully, I'll have a little while before I need to do that - and I'll be practicing between now and then! If you have any suggestions for new teachers to make concepts like these clearer and easier, please let me know!
He literally saved my life
god has a special place in heaven for you
This was such a wonderfully concise, easy to understand video! Thank youuuu 🙏
the love I have for this man <333333
am very happy with your video and when i will be done with this video i will be fucking genius
What about hcl ,how it dissociats in water and form ions even though it's a covalent compound
I HATE CHEMISTRY GRGRGRG... I'LL BE TAKING UPCAT THIS AUGUST AND ARGHHHHH HOW CAN I SOLVE THIS WITHOUT A CALCULATOR?! HUH!?!!
after 11 years this video is still pretty relevant ngl
Wow 👏👏👏👏
I’m preparing for high school and I forgot how to balance equations but after watching this I know how to balance
You’re nothing but the best ❤❤
Ok i got the best channel for This chapter thanku god 🙏💖
Reductor does not gain electrons, I think you confused this or I could have just interpeted this wrong 😂
Anyone here 2025
Wow!!
very helpful tysm
Holy this is helpful
21 zeroes after the number is a heptillion or a sextillion according to Google. Numbers names changed recently trillions changed into billions and the whole system got confused. It is still a great video though.
PLS TYLER COMEBACK
be m y teacher pls
Great explanation. I was with you up until the talk about the electrons being smaller than atoms. How were they able to make that assumption?
I was struggling and didn't understand how it works when I watched this video. Thank you!
Why didnt we directly write it as H + Cl = HCL
He makes everything easy
I love you and watch your videos after 9 and 12 years , this is sooo crazy😂!!! and I don’t know even if you still on RUclips and if you will read my comment or not , you really really helped me I just want to say thank uuuuu🥹🤍🤍🤍🤍🤍🤍🤍🤍🤍🤍🤍🤍🤍🤍🤍
"Thank you. This really helped me better than my teacher's explanation."
I scanned the QR code in my book and reached here. I'm not going backkkkkk! This is too good 🤩
What book is that?!? I had no idea my video was in a book!
Thanks!!!
Wow! Thank you so much!! It is very helpful video for me.
Thank you so much man
You are the best
1:49 bro went out of context
I keep getting confused by the coefficients and subscripts. Why isn't the multiplied oxidation number always the oxidation number? Like 2NO. O=-2 and not -4, but, N = +4 to equal 0. My brain wants to make O=-4 and not -2.
YOU. ARE. AMAZZIIINNGGG!
One of the best videos I have ever seen in my life
Thanks for your effort and for your excellent explanation ❤❤❤
i dont get why they have different number of protons than electrons. I assumed we started with neutral charged atoms which means they have same protons as electrons right? so in NH4+ how did you know there were only 10 electrons if there are 11 protons?
Dont think he explained why Copper has a stronger pull of electrons than zinc. Anyone knows why?
I have a chemistry test in an hour and I’m here learning what limiting reagents are.
Isn't the first example butene not ethene?
This video should get much more play than it does. It's one of the better ones on this subject on RUclips. The other ones I found were more like watching people do their homework.
Best teacher so far for me 😊
Who's here in 2024 😂 Then 1 year later some one will say who's here in 2025 😂❤
I have a regents in 30 minutes
this is more clear than any 3d animations or presentation